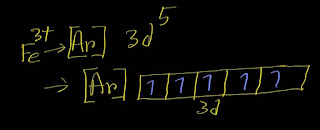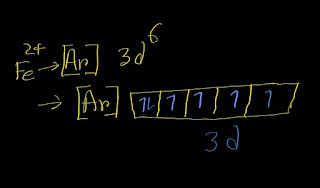In the ground-state electron configuration of iron, Fe, four unpaired electrons are present. Explanation: To find out the number of unpaired electrons in Fe, you need to write the electron configuration of Fe. Remember that the atomic number of iron is 26. Here is the ground state electron configuration of Fe: [Ar]3d⁶4s² Remember that the d subshell has five orbitals.Each orbit can take maximum two electrons. In case of filling degenerate orbits ,each orbital will take one electron before pairing by the second electron according to Hunds' rule. Next In the ground-state electron configuration of Fe2+, four unpaired electrons are present. Explanation: To find out the number of unpaired electrons in Fe^2+ ion, you need to write the electron configuration of Fe^2+. Remember that the atomic number of iron is 26. Here is the electron configuration of Fe^2+: [Ar]3d⁶ Remember that the d subshell has five orbitals.Each orbit can take maximum two...