【 】 1s2 2s2 2p4 ||What element has the electron configuration of 1s2 2s2 2p4?
What is 1s2 2s2 2p4 ?
1s2 2s2 2p4 means that
●s subshell of first orbit or energy level has two electrons,
●s subshell of second orbit or energy level has two electrons,
●p subshell of second orbit or energy level has four electrons,
From 1s2 2s2 2p4 electron configuration,we get 2+2+4 = 8 electrons.
We know that electron's number is equal to proton number or atomic number for neutral atom.
Since here there is no words about charge,we can say that 1s2 2s2 2p4 indicates the element whose atomic number is 8.
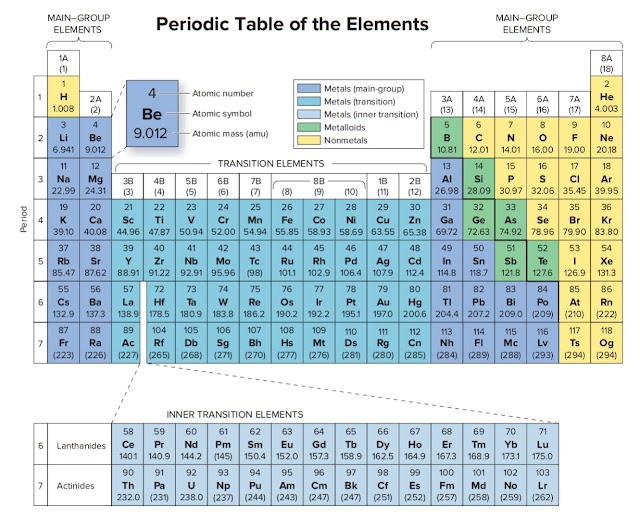
Now if you look at the periodic table, Oxygen has atomic number 8.






Comments
Post a Comment